FDA Food Facility Registration, U.S. Agent Representation & D-U-N-S® Registration: The Complete Guide for U.S. Market Entry

Entering the U.S. food market requires more than just a quality product—it requires proper facility identification, registration, and regulatory coordination. Before any food product can be legally imported or distributed in the United States, businesses must complete key administrative requirements, including:
- FDA Food Facility Registration
- U.S. Agent Representation (for foreign facilities)
- D-U-N-S® Number Registration
These are foundational steps for FDA compliance, yet many companies misunderstand their purpose, scope, and limitations.
This guide explains everything you need to know about FDA facility registration requirements, how to complete them correctly, and how to avoid common mistakes that delay market entry.
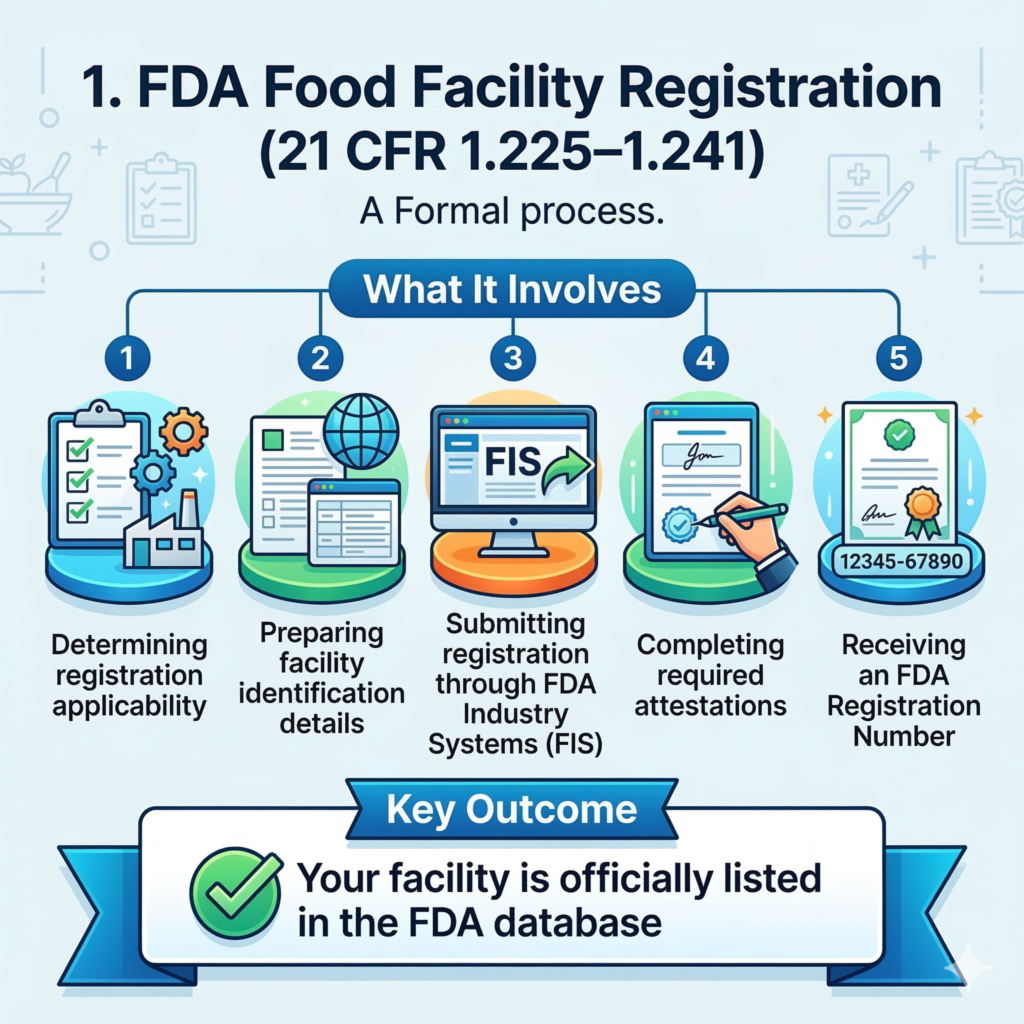
What Is FDA Food Facility Registration?
FDA Food Facility Registration is a mandatory requirement under the Food Safety Modernization Act (FSMA) and 21 CFR 1.225–1.241.
Any facility that:
- Manufactures
- Processes
- Packs
- Holds food for consumption in the United States must register with the FDA.
This applies to:
- U.S.-based facilities
- Foreign food facilities exporting to the U.S.
Without proper FDA facility registration, products may be:
- Refused entry at U.S. ports
- Subject to detention
- Flagged for regulatory review
Why FDA Registration Is Critical for Market Entry
Completing your FDA Food Facility Registration is not optional—it is a prerequisite for doing business in the U.S. food industry.
It enables:
- Facility identification within FDA systems
- Regulatory communication between FDA and the facility
- Traceability in case of safety events
However, it’s important to understand:
- FDA registration does NOT mean product approval.
- It does NOT replace compliance programs like FSVP or HACCP.
It is simply the first step in regulatory visibility and accountability. At FSVPServices.com, we often see businesses confuse registration with compliance—leading to significant gaps later.
Understanding the Three Core Requirements


Common Misconceptions About FDA Registration
Myth #1: FDA Registration = Approval
Incorrect – Registration only identifies your facility; it does not approve your product.
Myth #2: Registration Covers Compliance
Incorrect – You still need:
- FSVP (for importers)
- HACCP / Preventive Controls
- Labeling compliance
Myth #3: U.S. Agent Handles Everything
Incorrect – The U.S. Agent only facilitates communication between your facility and the FDA.
Scope of Administrative Registration Services
Our FDA Facility Registration + U.S. Agent + D-U-N-S® setup service is designed to provide administrative facilitation only.
FDA Food Facility Registration Assistance
- Review of client-provided information
- Submission facilitation via FDA Industry Systems
- Assistance with required attestations
- Delivery of registration confirmation
- Optional: Renewal or update facilitation
U.S. Agent Representation
- Acceptance of designation
- Provision of U.S. contact details
- Receipt and forwarding of FDA communications
- Coordination of inspection notices
- Includes limited communication support (standard hours annually)
D-U-N-S® Registration Support
- Record search and validation
- Data alignment assistance
- Registration facilitation (if required)
- Confirmation tracking
Administrative Coordination
- Alignment of facility data across systems
- Delivery of confirmation records
- Guidance on maintaining records
Important Compliance Disclaimer
- This service is strictly administrative, facilitative, and based on client-provided information
- It does NOT include regulatory consulting, compliance management, legal advisory, or FDA approval
- Clients remain fully responsible for regulatory compliance, responding to FDA findings, and maintaining food safety systems
Deliverables You Can Expect
- FDA Registration Submission Confirmation
- FDA Registration Number (when issued)
- U.S. Agent Designation Confirmation
- Forwarded FDA Communications (if received)
- D-U-N-S® Validation or Confirmation (if issued)
Why Structured Registration Matters
Potential Issues from Errors in FDA Facility Registration:
- Delayed approvals
- Incorrect facility records
- Communication breakdowns
- Import issues
A Structured Approach Ensures:
- Accuracy
- Consistency
- Proper documentation
At FSVPServices.com, we emphasize building foundational regulatory infrastructure that supports long-term compliance—not just one-time submissions.
Why Cosmetic Label Compliance Matters
Imported cosmetics must show country of origin (19 CFR §134) for customs clearance, import compliance, and consumer transparency. Failing to comply = delayed shipments and regulatory headaches.
Compliance isn’t a one-time task—it’s a system:
- Continuous monitoring
- Structured processes
- Integrated systems
At FSVPServices.com, we go beyond checklists, building compliance frameworks that include:
- Document control
- Supplier management
- Risk assessment
- Corrective actions
- Training & competency
Your label becomes more than just packaging—it’s your first impression, regulatory defense, and market entry tool.

Who Needs This Service
Who Needs This Service
- Foreign food manufacturers exporting to the U.S.
- Importers setting up supplier facilities
- Startups entering the U.S. food market
- Companies lacking a U.S. Agent
- Businesses requiring D-U-N-S® registration
Common Challenges We Solve
- Understanding FDA registration requirements
- Incorrect facility data submission
- Missing U.S. Agent designation
- D-U-N-S® inconsistencies
- Lack of coordination across systems
Our structured process eliminates these issues.
The Advantage of a System-Driven Approach
- Align registration with compliance systems
- Maintain consistent records
- Scale across multiple facilities
- Reduce regulatory risk
Even for administrative services, structure matters. Platforms like FSVPServices.com help ensure long-term efficiency and compliance readiness.
Frequently Asked Questions
Is FDA registration required for all food facilities?
Yes, if the facility manufactures, processes, packs, or holds food for U.S. consumption.
Does FDA registration mean my product is approved?
No. It only identifies your facility.
Do foreign facilities need a U.S. Agent?
Yes, it is mandatory.
Is a D-U-N-S® number always required?
In many cases, yes—for identification and alignment purposes.
Final Thought: Start with the Right Foundation
FDA registration is your entry point into the U.S. market.
Done correctly, it creates:
- Clear regulatory identity
- Proper communication channels
- Strong operational foundation
Done incorrectly, it creates delays, confusion, and risk.
At FSVPServices.com, we help businesses establish this foundation correctly—so they can build full compliance systems with confidence.
Start Your FDA Registration Process Today
If you need support with:
- ✅ FDA Food Facility Registration
- ✅ U.S. Agent Representation
- ✅ D-U-N-S® Registration
Final Thought
Reach out today to begin your FDA registration setup and ensure full compliance.
Simplified FDA Registration. Confident Compliance. Peace of Mind.










